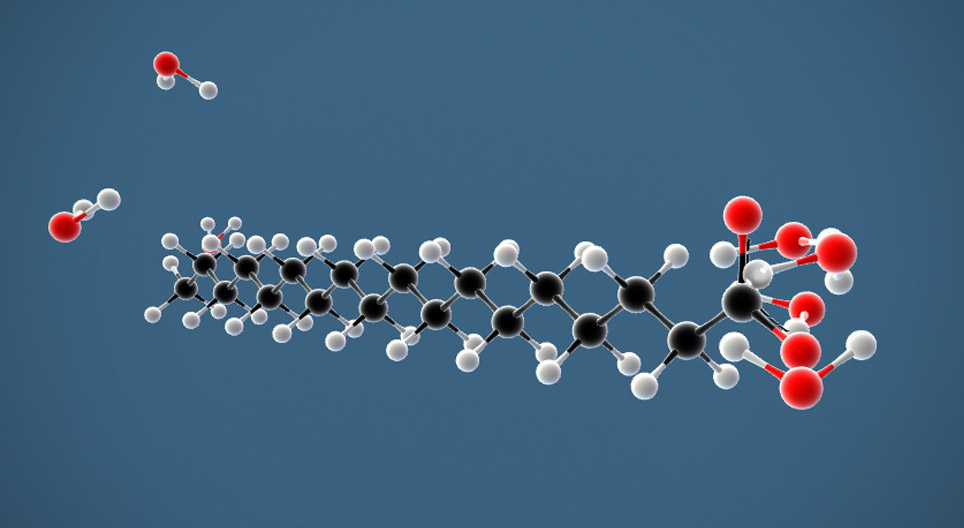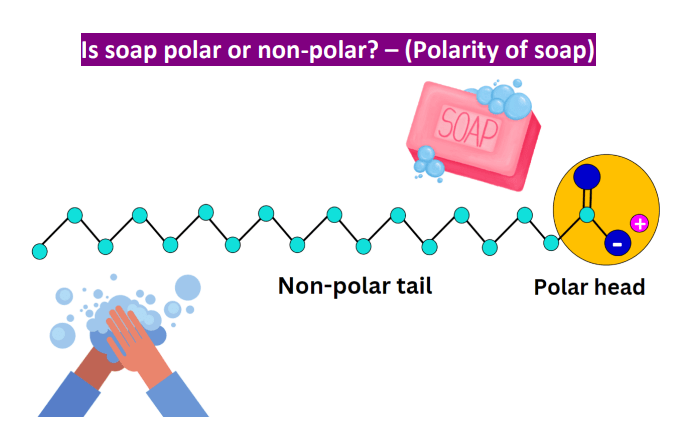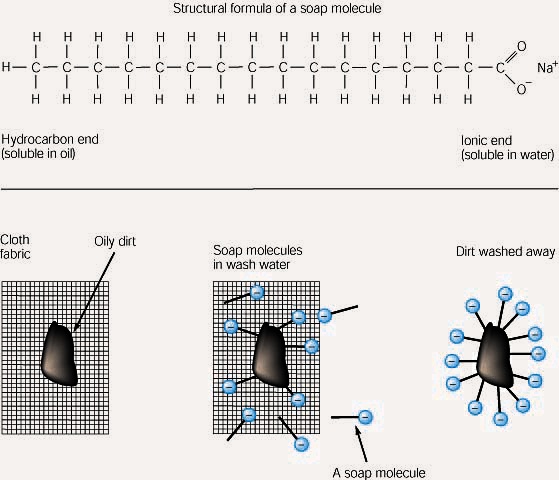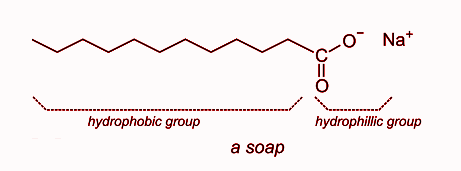
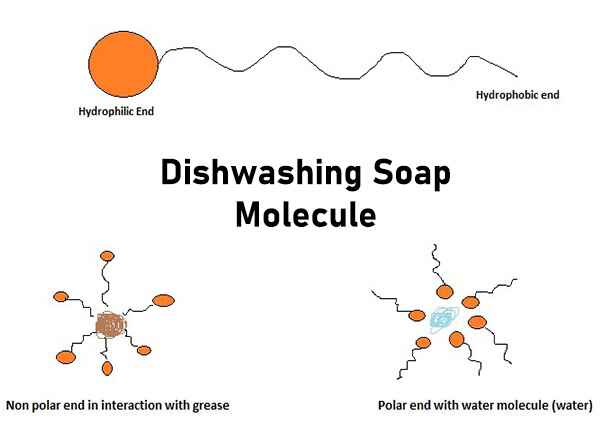
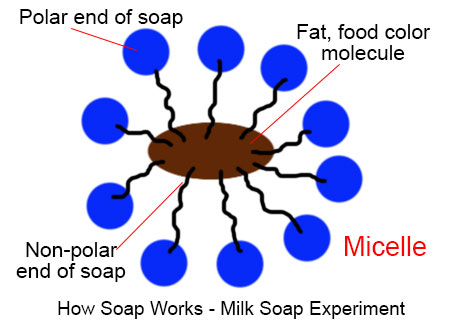
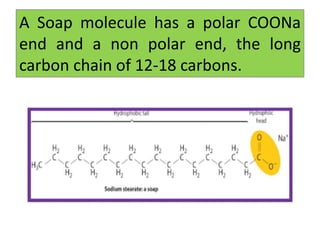
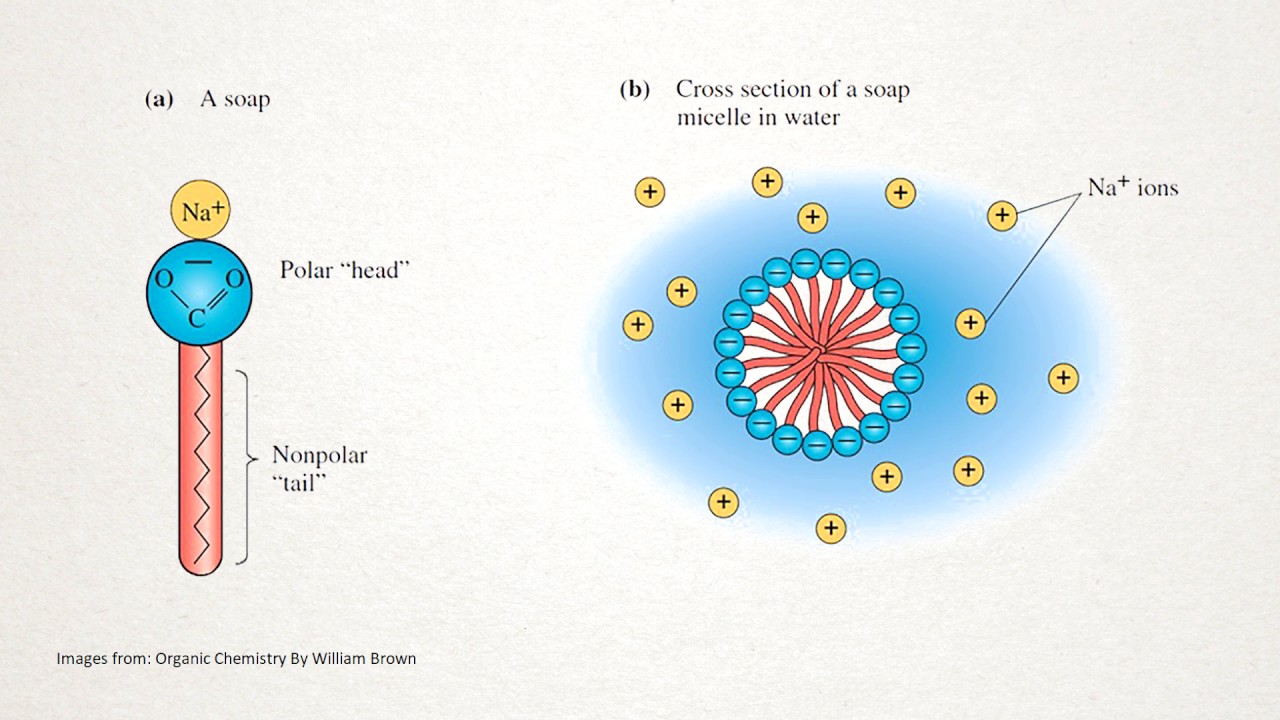
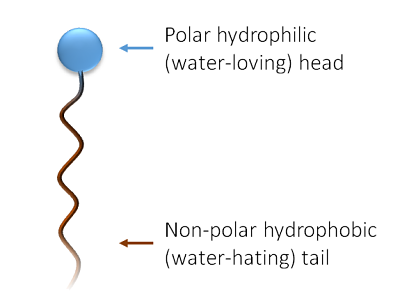
a) What is soap? (b) Describe the structure of a soap molecule with the help of a diagram. (c) Explain the cleansing action of soap. Draw a diagram to illustrate your answer.

TRUE OR FALSE? Micelles form to keep the long non-polar tail of the soap molecule away from water. | Homework.Study.com
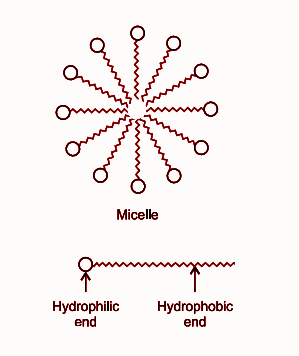
a) What is soap? (b) Describe the structure of a soap molecule with the help of a diagram. (c) Explain the cleansing action of soap. Draw a diagram to illustrate your answer.

Why does micelle formation take place when soap is added to water? Will a micelle be formed in other solvents such as ethanol also?






:max_bytes(150000):strip_icc()/soap-micelle-58ed36193df78cd3fcdf0908.jpg)