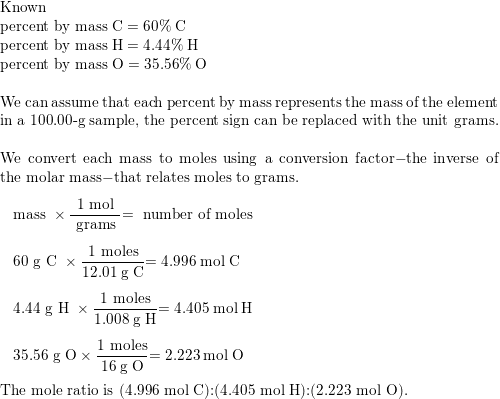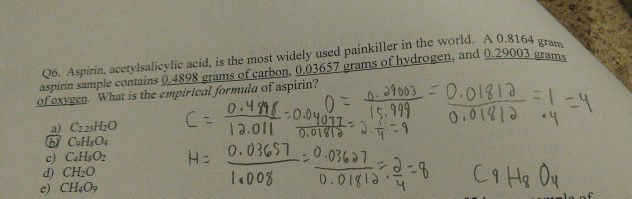Arrange the proper sequence of the steps to determine the refractive index of a prism by using angle of minimum deviation method(a) Fix a white sheet of paper on a drawing board.(b)
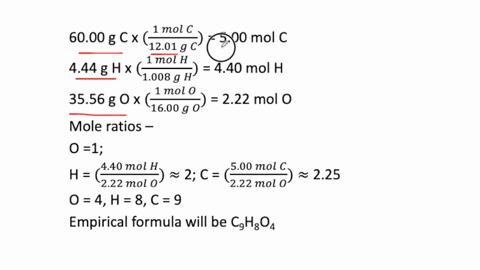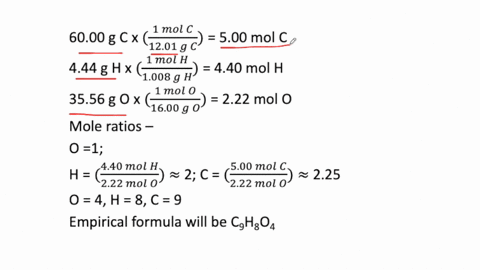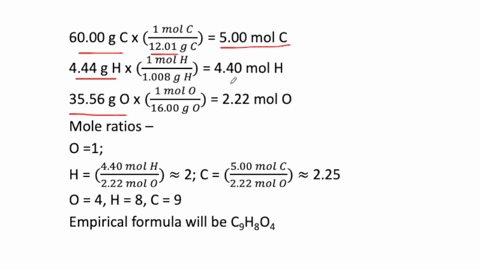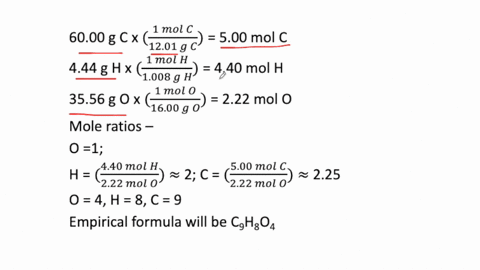
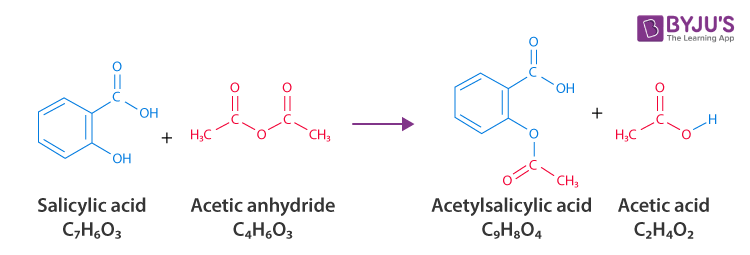
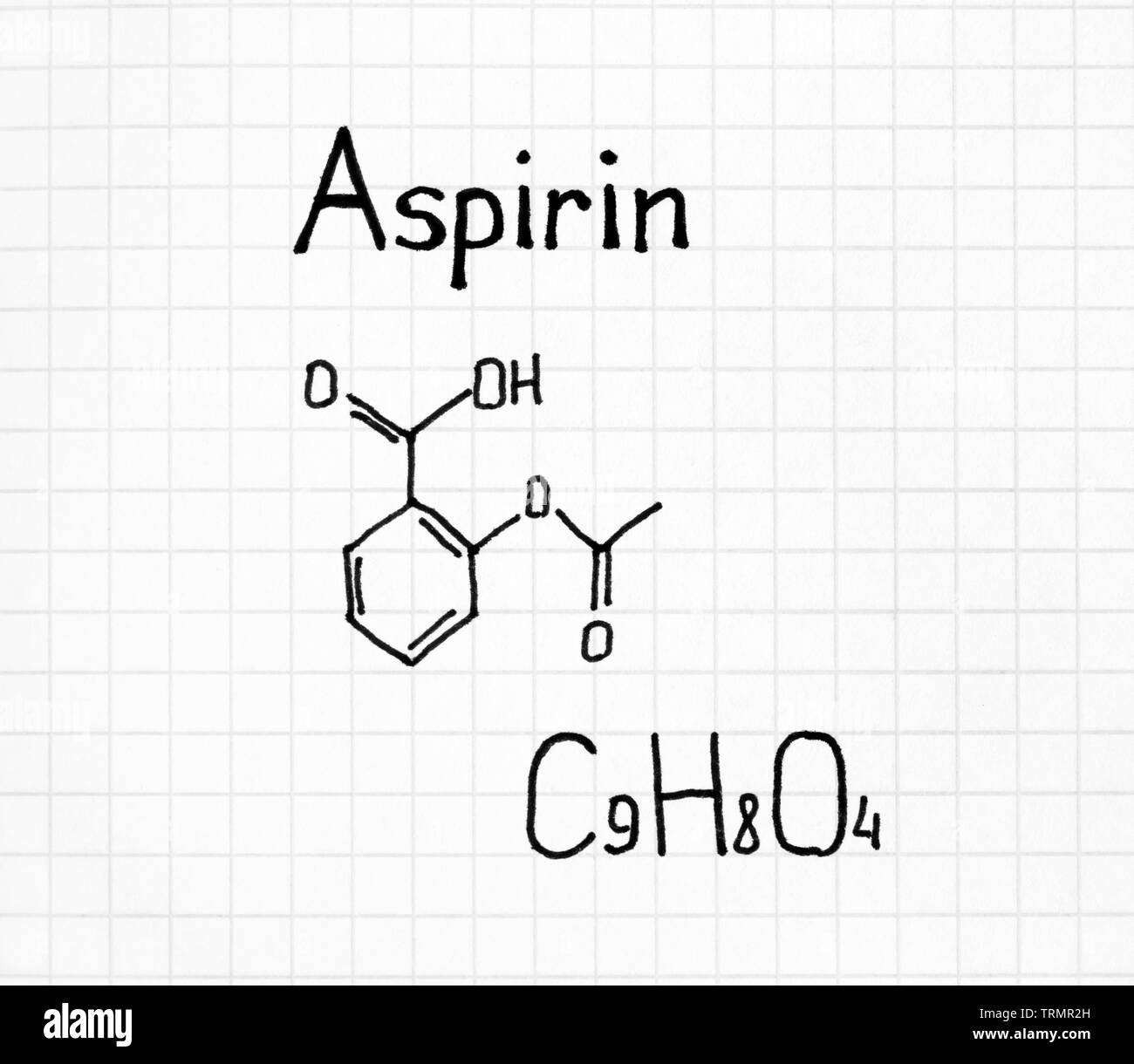
SOLVED: Determine the empirical formula for 1.2 grams of aspirin. It contains 60.0% C, 4.44% H and 35.56% 0

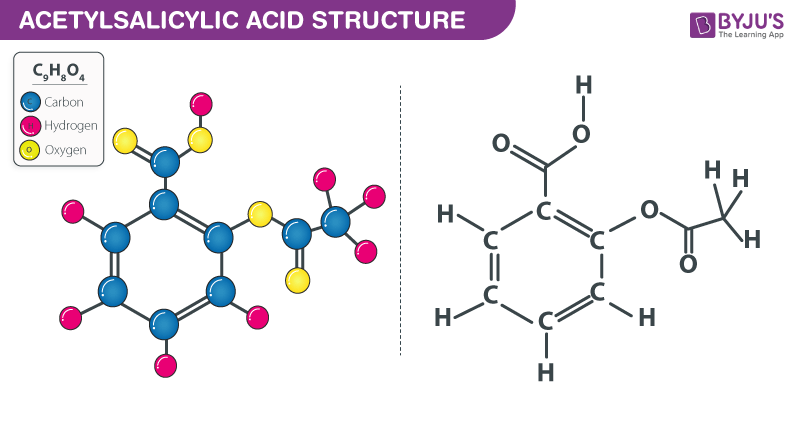
Aspirin has a molar mass of 180g/mol. If the empirical formula is C9H8O4, what is the molecular formula of - brainly.com

SOLVED: A laboratory analysis of aspirin determined the following mass percent composition: C 60.00%, H 4.48%, O 35.52%. Find the empirical formula. ****I KNOW THE ANSWER FOR THIS IS C9H804, BUT I